
In my 10 years as a medical executive in health tech start-ups, I noticed that medical/science people do not necessarily appreciate the difference between the different types of engineers or designers. To be honest, when I came to Silicon Valley, I considered all people who do anything with computers to simply be “IT people.” Likewise, many people from the tech industry don’t necessarily know the difference between the different types of scientists or the different types of medical doctors—or even the difference between a medical doctor (MD) and a research scientist (PhD).
Good Science Doesn’t Instantly Translate into a Viable Health Product
Tech investors are increasingly interested in the $4-trillion US healthcare industry. Many are new to this sector. Although they use lawyers to do their legal diligence, they often use their own associates to do their “medical diligence.” Understanding the difference between medical training and scientist training helps to explain why the passion of a scientist entrepreneur may not necessarily translate into a viable health product or why the associate who did your medical diligence missed all the red flags that blocked your investment from widespread market adoption.
Scientific validity is not the same as clinical validity, yet most of the literature provides scientific validity. In addition, not all MDs are the same. Some specialties get more training in clinical trials, while others develop more expertise in digital imaging, informatics, or device/test development. A dermatologist MD may not be the best person to handle medical diligence for a home laboratory testing start-up.
Medical Review Isn’t the Same as Scientific Review
The confusion about medical and scientific training can impact efficiency in health tech startups. Once, after working at a particular startup for a couple years, I got frustrated at the commercial team for not getting medical review before releasing new collateral, despite multiple requests. When we met to talk about it, they explained that they did have medical review because a PhD scientist had approved the collateral. I said, “It’s good to get a scientific review, but it’s not the same as a medical review.” One of my teammates asked, “What’s the difference?” When I explained it, many of them seemed genuinely surprised. The purpose of this blog is to shorten the learning cycle on that epiphany.
(In a future blog, I’ll try to explain the different kinds of engineers to the healthcare industry. And I’ll tell them not to call software engineers “IT people.” I will also explain the difference between scientific and clinical validity, and what is required to bridge the gap between interesting science and a viable medical product.)

Table 1: The training and expertise of medical doctors versus research scientists.
Not All MDs Are the Same
The next table addresses the high-level differences between the different kinds of MDs. This classic joke sums up the stereotypes quite nicely:
“A medical student, an internist, a radiologist, a surgeon and a pathologist go duck hunting. They barely find their duck blind before the first duck flies over. The medical student is the first to raise his shotgun, but unable to tell if the duck is really a duck, he does not shoot. The internist aims his shotgun, but can not tell for sure what subspecies of duck it is and wants to order more tests. He does not shoot. He asks the radiologist to take a picture of the duck. The surgeon quickly raises his shotgun, aims, and without pause shoots. The duck falls to the ground. The surgeon turns to the pathologist. “Go figure out if that’s a duck or not.” Funny, right? Even other doctors don’t really know what pathologists do. Pretty sure my family doesn’t. Now I can refer them to this blog.
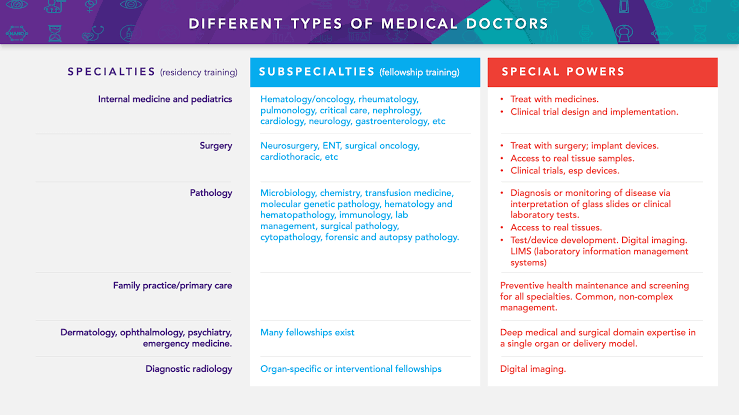
Table 2: Different types of medical doctors
Not All Lab Directors Are Created Equal
Some health tech companies choose to operate their own clinical laboratories, if their product includes a clinical test. The decision to operate one’s own lab rather than outsource the testing to an established clinical testing laboratory has important pros and cons that will be unpacked in a future blog. But one of the requirements of a clinical laboratory is that it is overseen by appropriately credentialed (and in some states, licensed) clinical laboratory professionals. So it is relevant to this blog to know who those professionals might be.
Most types of clinical testing must be overseen and interpreted by a MD. However, a few types of clinical tests can be overseen by credentialed PhDs who have done a post-doctoral clinical laboratory fellowship. These include germ-line genetic testing, microbiology, chemistry, and toxicology. For many tech people, the difference between MD pathologists (who are trained to run all types of clinical labs) and subspecialty-focused clinical PhDs is not obvious. Using the example of genetics, the following table outlines the difference between the MDs and the PhDs.

Table 3: Different types of laboratory directors allowed by CLIA ‘88 regulations.
The bottom line: Know your experts and what each is best suited for. Tapping the precise expertise you need will get your health product faster into the hands of the people who need it.